PtsI independently regulates cell length and width through multiple metabolic and signaling pathways
Abstract
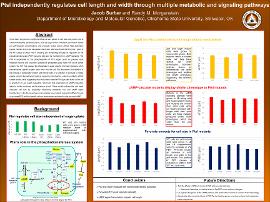
It has been long known that Escherichia coli can adjust its cell size and growth rate to nutrient availability. Several proteins, such as OpgH (which modulates cell division based on UDP-glucose concentration), and pyruvate kinase (which affects FtsZ assembly) couple nutrition to the cell elongation machinery. We have found that Enzyme I (ptsI) of the PTS plays a critical role in dictating cell morphology through its regulation of the phosphoenolpyruvate (PEP):pyruvate ratio and its involvement in cAMP signaling. The PTS is responsible for the phosphorylation of PTS sugars (such as glucose) upon entrance into the cell; Enzyme I passes the phosphate group from PEP to the carrier protein Hpr. HPr then passes the phosphate to sugar specific Enzyme II proteins which phosphorylate specific sugars upon entry into the cell. We discovered that deletion of ptsI results in significantly shorter and thinner cells. The addition of pyruvate to media greatly corrects the cell length defects caused by ptsI deletion, while the addition of PEP further shrinks cells, suggesting that PtsI control over the PEP:pyruvate levels in the cell is important for cell length regulation. We found that interruption of cAMP production leads to similar outcomes as the absence of ptsI. These results indicate that ptsI might modulate cell size by separately influencing metabolite flux and cAMP signal transduction. In the future we hope to investigate how mutants respond to different levels of pyruvate/PEP and the specific cellular mechanisms activated by ptsI-induced cAMP.
Citation
Surber, J., & Morgenstein, R. (2023, April 18). PtsI independently regulates cell length and width through multiple metabolic and signaling pathways. Poster session presented at the Oklahoma State University Undergraduate Research Symposium, Stillwater, OK.